The amount of paramagnetism for a first-series transition metal complex is related approximately to its spin-only magnetic moment. The spin-only value of the magnetic moment in units of Bohr magnetons (BM) is given by sqrt(n(n + 2)), where n is the number of unpaired electrons. Calculate the spin-only value of the magnetic moment for the 2+ ions of the first-series transition metals (except Sc) in octahedral complexes with (a) weak-field ligands and (b) strong-field ligands. For which electron configurations can the magnetic moment distinguish between high-spin and low-spin electron configurations?
Ch.21 - Transition Elements and Coordination Chemistry

All textbooks McMurry 8th Edition
McMurry 8th Edition Ch.21 - Transition Elements and Coordination Chemistry
Ch.21 - Transition Elements and Coordination Chemistry Problem 21.106
Problem 21.106
 McMurry 8th Edition
McMurry 8th Edition Ch.21 - Transition Elements and Coordination Chemistry
Ch.21 - Transition Elements and Coordination Chemistry Problem 21.106
Problem 21.106Chapter 21, Problem 21.106
For each of the following complexes, draw a crystal field energy-level diagram, assign the electrons to orbitals, and predict the number of unpaired electrons.
(a) [CrF6]3-
(b) [V(H2O)6]3+
(c) [Fe(CN)6]3-
 Verified step by step guidance
Verified step by step guidance1
Identify the oxidation state of the metal in each complex and determine the electron configuration of the metal ion.
Determine the geometry of each complex (octahedral, tetrahedral, etc.) and use it to draw the crystal field splitting diagram.
Assign the electrons to the d-orbitals according to the crystal field splitting diagram, considering the electron configuration and the nature of the ligands (strong or weak field).
Count the number of unpaired electrons in the d-orbitals for each complex.
Use the number of unpaired electrons to predict the magnetic properties of each complex (paramagnetic or diamagnetic).

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Was this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Crystal Field Theory
Crystal Field Theory (CFT) explains how the arrangement of ligands around a central metal ion affects the energy levels of the d-orbitals. In an octahedral complex, for example, the d-orbitals split into two energy levels: the lower-energy t2g and the higher-energy eg orbitals. This splitting is crucial for determining the electronic configuration of the metal ion and predicting properties such as color and magnetism.
Recommended video:
Guided course

The study of ligand-metal interactions helped to form Ligand Field Theory which combines CFT with MO Theory.
Electron Configuration and Orbital Filling
The electron configuration of a transition metal ion is determined by the number of electrons it has after accounting for its oxidation state. Electrons fill the d-orbitals according to the Aufbau principle, Hund's rule, and the Pauli exclusion principle. Understanding how to assign electrons to the split d-orbitals in a crystal field diagram is essential for predicting the number of unpaired electrons, which influences the magnetic properties of the complex.
Recommended video:
Guided course

Electron Orbital Diagrams
Ligand Field Strength and Crystal Field Splitting
The strength of the ligands surrounding a metal ion affects the degree of d-orbital splitting, known as crystal field splitting energy (Δ). Strong field ligands, like CN⁻, cause a larger splitting, leading to lower energy configurations and potentially fewer unpaired electrons. Conversely, weak field ligands, like F⁻, result in smaller splitting, which can lead to more unpaired electrons. This concept is vital for predicting the magnetic behavior of the complexes.
Recommended video:
Guided course
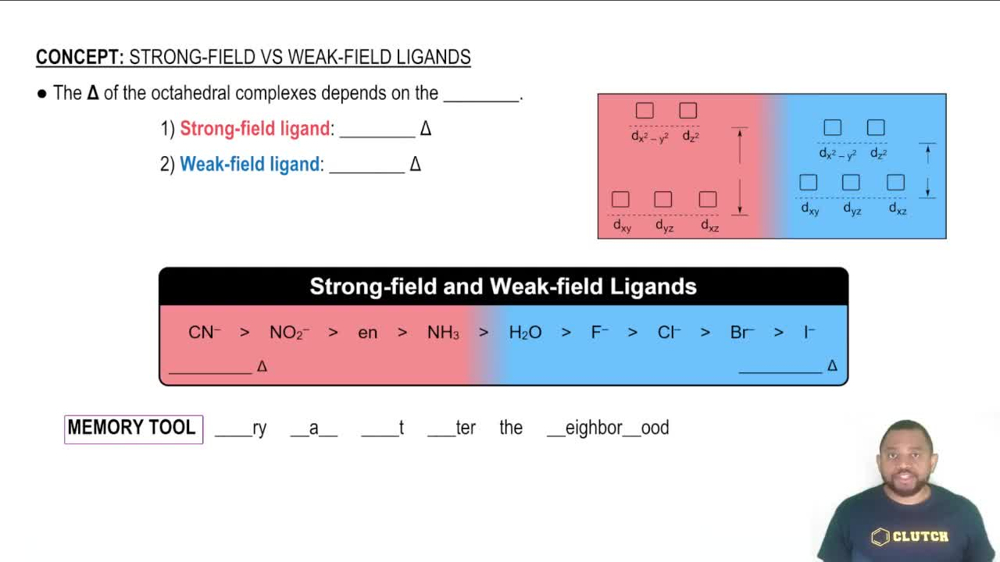
Strong-Field Ligands result in a large Δ and Weak-Field Ligands result in a small Δ.
Related Practice
Textbook Question
99
views
Textbook Question
What is the systematic name for each of the following coordination compounds?
(c) [Co(NH3)4Br2]Br
(d) Cu(gly)2
170
views
Textbook Question
What is the name of the compound [Fe(H2O)5(SCN)]Cl2?
(a) pentaaquathiocyanatoiron(III) chloride
(b) pentaaquachlorothiocyanato iron(III)
(c) pentaaquathiocyanatoiron(III) dichloride
(d) pentaaquathiocyanatoiron(II) chloride
491
views
Textbook Question
The Ni2+(aq) cation is green, but Zn2+(aq) is colorless. Explain.
96
views
Textbook Question
Draw the structure of all isomers of the octahedral complex [NbX2Cl4]- (X- = NCS-), and identify those that are linkage isomers.
99
views
Textbook Question
Which of the following complexes can exist as enantiomers? Draw their structures.
(a) [Cr(en)3]3+
(b) cis-[Co(NH3)Cl]2+
(c) trans-[Co(en)2(NH3)Cl]2+
(d) [Pt(NH3)3Cl3]+
112
views