What is the highest oxidation state for each of the elements from Sc to Zn?

Which of the following complexes can exist as enantiomers? Draw their structures.
(a) [Cr(en)3]3+
(b) cis-[Co(NH3)Cl]2+
(c) trans-[Co(en)2(NH3)Cl]2+
(d) [Pt(NH3)3Cl3]+
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
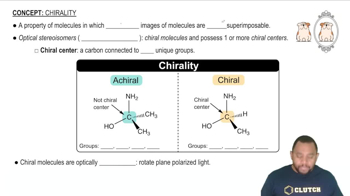
Chirality
Coordination Complexes
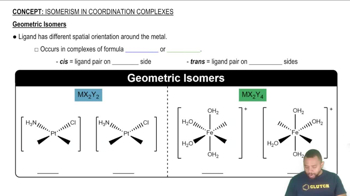
Geometric Isomerism
What is the oxidation state of the metal in each of the complexes?
a. AgCl2–
b. [Cr(H2O)5Cl]2+
c. [Co(NCS)4]2–
d. [ZrF8]4–
e. [Fe(EDTA)(H2O)]–
What role does EDTA4- play as a trace additive to mayonnaise? Would the glycinate ion (H2NCH2CH2NH2) be an effective substitute for EDTA4-?
Based on the wavelength of maximum absorption of the cobalt complexes, arrange the following ligands in a spectrochemical series from weakest-field to strongest-field ligand.
(a) Cl- < NCS- < H2O < NH3
(b) Cl- < NCS- < H2O < NH3
(c) H2O < Cl- < NH3 < NCS-
(d) Cl- < H2O < NCS- < NH3
What hybrid orbitals are used by the metal ion and how many unpaired electrons are present the complex ion [VCl4]- with tetrahedral geometry?
(a) sp3; 2 unpaired electrons
(b) sp3; 3 unpaired electrons
(c) sp3d2; 3 unpaired electrons
(d) sp3d2; 4 unpaired electrons
Predict the number of unpaired electrons for each of the following.
(a) Sc3+
(b) Co2+
