Draw a crystal field energy-level diagram for a square planar complex, and explain why square planar geometry is especially common for d8 complexes.

 McMurry 8th Edition
McMurry 8th Edition Ch.21 - Transition Elements and Coordination Chemistry
Ch.21 - Transition Elements and Coordination Chemistry Problem 21.115
Problem 21.115Which of the following complexes are diamagnetic?
(a) [Ni(H2O)6]2+
(b) [Co(CN)6]3-
(c) [HgI4]2- (tetrahedral)
(d) [Cu(NH3)4]2+ (square planar)
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Magnetism in Coordination Complexes
Crystal Field Theory

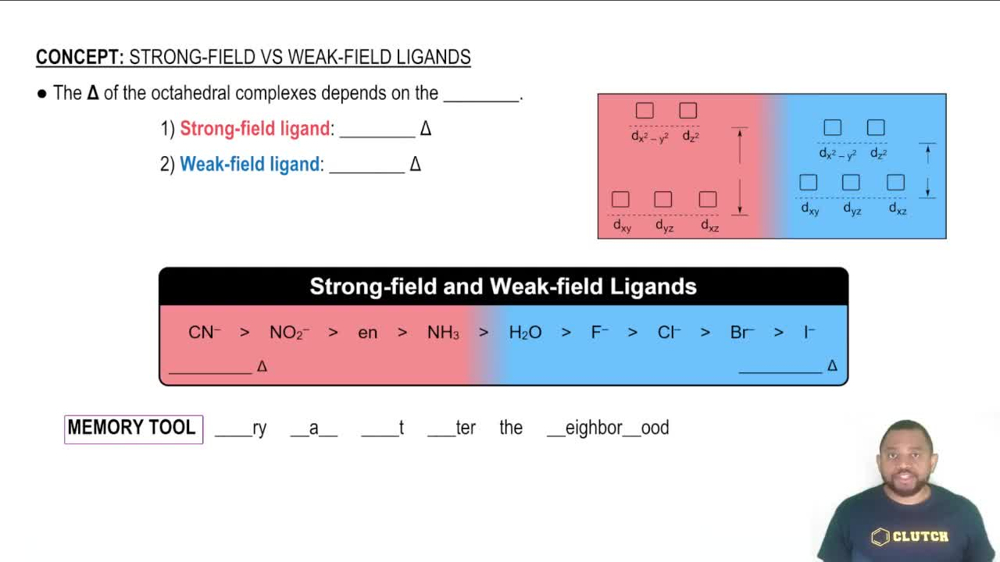
Ligand Field Strength
Predict the crystal field energy-level diagram for a square pyramidal ML5 complex that has two ligands along the axes but only one ligand along the z axis. Your diagram should be intermediate between those for an octahedral ML6 complex and a square planar ML4 complex.
What is a racemic mixture? Does it affect plane-polarized light? Explain.
The amount of paramagnetism for a first-series transition metal complex is related approximately to its spin-only magnetic moment. The spin-only value of the magnetic moment in units of Bohr magnetons (BM) is given by sqrt(n(n + 2)), where n is the number of unpaired electrons. Calculate the spin-only value of the magnetic moment for the 2+ ions of the first-series transition metals (except Sc) in octahedral complexes with (a) weak-field ligands and (b) strong-field ligands. For which electron configurations can the magnetic moment distinguish between high-spin and low-spin electron configurations?
What is the systematic name for each of the following coordination compounds?
(c) [Co(NH3)4Br2]Br
(d) Cu(gly)2
What is the name of the compound [Fe(H2O)5(SCN)]Cl2?
(a) pentaaquathiocyanatoiron(III) chloride
(b) pentaaquachlorothiocyanato iron(III)
(c) pentaaquathiocyanatoiron(III) dichloride
(d) pentaaquathiocyanatoiron(II) chloride