The following pictures represent structures of the hydrides of four second-row elements:
(1)
(2)
(3)
(4)
(a) Which compound has the highest melting point?

 Verified step by step guidance
Verified step by step guidance

The following pictures represent structures of the hydrides of four second-row elements:
(1)
(2)
(3)
(4)
(a) Which compound has the highest melting point?
Write a balanced net ionic equation for the reaction of the amphoteric oxide Ga2O3 with:
a. Aqueous sulfuric acid
An important physiological reaction of nitric oxide (NO) is its interaction with the superoxide ion (O2–) to form the peroxynitrite ion (ONOO–).
a. Write electron-dot structures for NO, O2–, and ONOO–, and predict the O–N–O bond angle in ONOO–.
In the following pictures of binary hydrides, ivory spheres
represent H atoms or ions, and burgundy spheres represent
atoms or ions of the other element.
(1)
(2)
(3)
(4)
(a) Identify each binary hydride as ionic, covalent, or interstitial.
(b) What is the oxidation state of hydrogen in compounds (1), (2), and (3)? What is the oxidation state of the other
element?
The following pictures represent various silicate anions. Write the formula and charge of each anion.
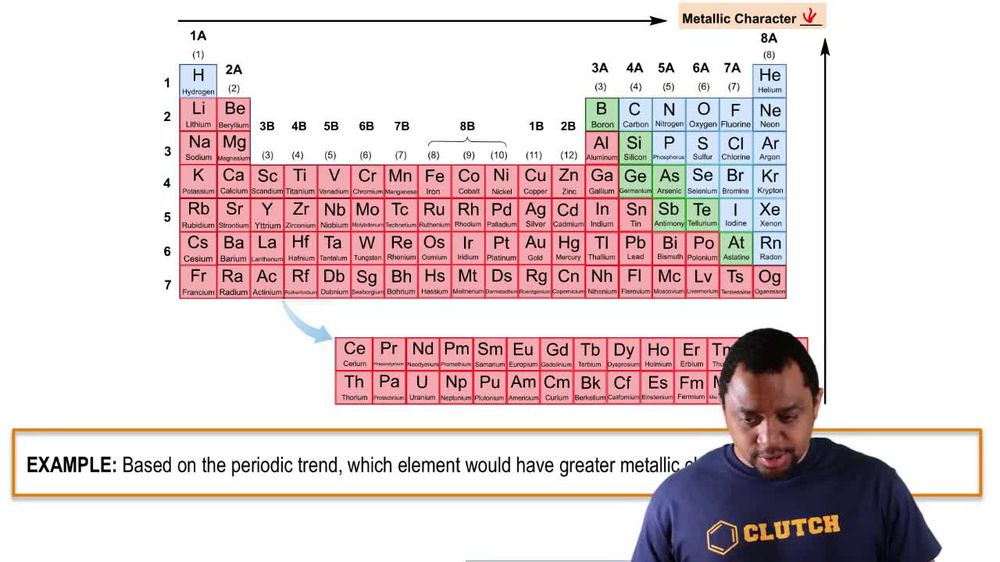
Consider the six second- and third-row elements in groups 4A–6A of the periodic table:
Possible structures for the binary fluorides of each of these elements in its highest oxidation state are shown below.
(b) Explain why the fluorides of nitrogen and phosphorus have different molecular structures but the fluorides of carbon and silicon have the same molecular structure.