Consider the chemical reaction:
C(s) + H2O(g) → CO(g) + H2(g)
How many liters of hydrogen gas are formed from the complete reaction of 22.5 g C? Assume that the hydrogen gas is collected at a pressure of 1.00 atm and a temperature of 355 K.

 Verified step by step guidance
Verified step by step guidance


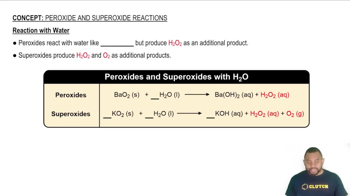
Consider the chemical reaction:
C(s) + H2O(g) → CO(g) + H2(g)
How many liters of hydrogen gas are formed from the complete reaction of 22.5 g C? Assume that the hydrogen gas is collected at a pressure of 1.00 atm and a temperature of 355 K.
A heliox deep-sea diving mixture contains 2.0 g of oxygen to every 98.0 g of helium. What is the partial pressure of oxygen when this mixture is delivered at a total pressure of 8.5 atm?
The hydrogen gas formed in a chemical reaction is collected over water at 30.0 °C at a total pressure of 732 mmHg. What is the partial pressure of the hydrogen gas collected in this way? If the total volume of gas collected is 722 mL, what mass of hydrogen gas is collected?
Consider the chemical reaction: 2 H2O(l) → 2 H2(g) + O2(g) What mass of H2O is required to form 1.85 L of O2 at a temperature of 315 K and a pressure of 0.981 atm?
What is the mole fraction of oxygen gas in air (see Table 6.3)? What volume of air contains 20.0 g of oxygen gas at 273 K and 1.00 atm?
The air in a bicycle tire is bubbled through water and collected at 25 °C. If the total volume of gas collected is 5.45 L at a temperature of 25 °C and a pressure of 745 torr, how many moles of gas were in the bicycle tire?