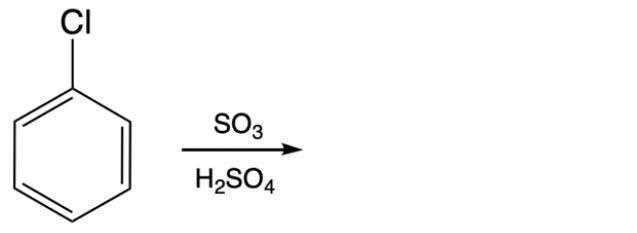
Sulfonation is a key electrophilic aromatic substitution reaction where benzene reacts with sulfur trioxide (SO3) in the presence of sulfuric acid (H2SO4) as a catalyst. This process involves replacing one hydrogen atom on the benzene ring with a sulfonic acid group (–SO3H). The sulfuric acid catalyst plays a crucial role by activating the sulfur atom in the sulfonic acid group, facilitating the substitution.
During the reaction, the sulfur trioxide provides the –SO3 portion, while the hydrogen atom incorporated into the sulfonic acid group originates from the sulfuric acid. The product formed is benzene sulfonic acid, characterized by the attachment of the –SO3H group to the aromatic ring. This transformation exemplifies how electrophilic aromatic substitution can introduce functional groups onto benzene, altering its chemical properties and reactivity.