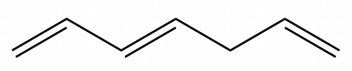
Dienes and trienes are organic compounds characterized by the presence of multiple carbon-carbon double bonds. Specifically, dienes contain two double bonds, while trienes have three. The nomenclature for these compounds follows rules similar to those used for naming alkanes, with key modifications to account for the multiple double bonds.
When naming dienes and trienes, the parent hydrocarbon chain is selected based on the longest continuous chain that includes the double bonds. The position of each double bond is indicated by the lowest possible numbers assigned to the carbon atoms involved. The suffix -diene is used for compounds with two double bonds, and -triene for those with three. Substituents attached to the main chain are named and numbered according to their position, maintaining the standard conventions of organic nomenclature.
For example, if a diene has double bonds starting at carbons 1 and 3 in a six-carbon chain, the compound would be named as a hexadiene with the numbers indicating the positions of the double bonds, such as 1,3-hexadiene. This systematic approach ensures clarity and consistency in naming complex unsaturated hydrocarbons.
Understanding the naming of dienes and trienes is essential for recognizing their structure and reactivity in organic chemistry, as the position and number of double bonds significantly influence their chemical behavior.