Alcohols can be named using common names, which is a straightforward method especially useful for simple alcohols. This naming convention involves stating the name of the alkyl group attached to the hydroxyl (–OH) group, followed by the word "alcohol." For example, an alcohol with a methyl group is called methyl alcohol, one with a propyl group is propyl alcohol, and one with a hexyl group is hexyl alcohol. This approach emphasizes the alkyl portion of the molecule first, making it easy to identify the structure based on the alkyl chain length and the presence of the alcohol functional group.
- 1. Matter and Measurements4h 29m
- What is Chemistry?7m
- The Scientific Method9m
- Classification of Matter16m
- States of Matter8m
- Physical & Chemical Changes19m
- Chemical Properties8m
- Physical Properties5m
- Intensive vs. Extensive Properties13m
- Temperature (Simplified)9m
- Scientific Notation13m
- SI Units (Simplified)5m
- Metric Prefixes24m
- Significant Figures (Simplified)11m
- Significant Figures: Precision in Measurements7m
- Significant Figures: In Calculations19m
- Conversion Factors (Simplified)15m
- Dimensional Analysis22m
- Density12m
- Specific Gravity9m
- Density of Geometric Objects19m
- Density of Non-Geometric Objects8m
- 2. Atoms and the Periodic Table5h 22m
- The Atom (Simplified)9m
- Subatomic Particles (Simplified)12m
- Isotopes17m
- Ions (Simplified)22m
- Atomic Mass (Simplified)18m
- Atomic Mass (Conceptual)12m
- Periodic Table: Element Symbols6m
- Periodic Table: Classifications11m
- Periodic Table: Group Names8m
- Periodic Table: Representative Elements & Transition Metals7m
- Periodic Table: Elemental Forms (Simplified)6m
- Periodic Table: Phases (Simplified)8m
- Law of Definite Proportions9m
- Atomic Theory9m
- Rutherford Gold Foil Experiment9m
- Wavelength and Frequency (Simplified)5m
- Electromagnetic Spectrum (Simplified)11m
- Bohr Model (Simplified)9m
- Emission Spectrum (Simplified)3m
- Electronic Structure4m
- Electronic Structure: Shells5m
- Electronic Structure: Subshells4m
- Electronic Structure: Orbitals11m
- Electronic Structure: Electron Spin3m
- Electronic Structure: Number of Electrons4m
- The Electron Configuration (Simplified)22m
- Electron Arrangements5m
- The Electron Configuration: Condensed4m
- The Electron Configuration: Exceptions (Simplified)12m
- Ions and the Octet Rule9m
- Ions and the Octet Rule (Simplified)8m
- Valence Electrons of Elements (Simplified)5m
- Lewis Dot Symbols (Simplified)7m
- Periodic Trend: Metallic Character4m
- Periodic Trend: Atomic Radius (Simplified)7m
- 3. Ionic Compounds2h 20m
- Periodic Table: Main Group Element Charges14m
- Periodic Table: Transition Metal Charges5m
- Periodic Trend: Ionic Radius (Simplified)5m
- Periodic Trend: Ranking Ionic Radii8m
- Periodic Trend: Ionization Energy (Simplified)9m
- Periodic Trend: Electron Affinity (Simplified)8m
- Ionic Bonding6m
- Naming Monoatomic Cations6m
- Naming Monoatomic Anions5m
- Polyatomic Ions25m
- Naming Ionic Compounds11m
- Writing Formula Units of Ionic Compounds7m
- Naming Ionic Hydrates6m
- Naming Acids18m
- 4. Molecular Compounds2h 18m
- Covalent Bonds6m
- Naming Binary Molecular Compounds6m
- Molecular Models4m
- Bonding Preferences6m
- Lewis Dot Structures: Neutral Compounds (Simplified)8m
- Multiple Bonds4m
- Multiple Bonds (Simplified)6m
- Lewis Dot Structures: Multiple Bonds10m
- Lewis Dot Structures: Ions (Simplified)8m
- Lewis Dot Structures: Exceptions (Simplified)12m
- Resonance Structures (Simplified)5m
- Valence Shell Electron Pair Repulsion Theory (Simplified)4m
- Electron Geometry (Simplified)8m
- Molecular Geometry (Simplified)11m
- Bond Angles (Simplified)11m
- Dipole Moment (Simplified)15m
- Molecular Polarity (Simplified)7m
- 5. Classification & Balancing of Chemical Reactions3h 17m
- Chemical Reaction: Chemical Change5m
- Law of Conservation of Mass5m
- Balancing Chemical Equations (Simplified)13m
- Solubility Rules16m
- Molecular Equations18m
- Types of Chemical Reactions12m
- Complete Ionic Equations18m
- Calculate Oxidation Numbers15m
- Redox Reactions17m
- Spontaneous Redox Reactions8m
- Balancing Redox Reactions: Acidic Solutions17m
- Balancing Redox Reactions: Basic Solutions17m
- Balancing Redox Reactions (Simplified)13m
- Galvanic Cell (Simplified)16m
- 6. Chemical Reactions & Quantities2h 34m
- 7. Energy, Rate and Equilibrium3h 45m
- Nature of Energy6m
- First Law of Thermodynamics7m
- Endothermic & Exothermic Reactions7m
- Bond Energy14m
- Thermochemical Equations12m
- Heat Capacity19m
- Thermal Equilibrium (Simplified)8m
- Hess's Law23m
- Rate of Reaction11m
- Energy Diagrams12m
- Rate Law (Simplified)5m
- Chemical Equilibrium7m
- The Equilibrium Constant14m
- Le Chatelier's Principle20m
- Solubility Product Constant (Ksp)17m
- Spontaneous vs Nonspontaneous Reactions7m
- Entropy (Simplified)9m
- Gibbs Free Energy (Simplified)18m
- 8. Gases, Liquids and Solids3h 27m
- Pressure Units6m
- Kinetic Molecular Theory14m
- The Ideal Gas Law18m
- The Ideal Gas Law Derivations13m
- The Ideal Gas Law Applications6m
- Chemistry Gas Laws17m
- Chemistry Gas Laws: Combined Gas Law12m
- Standard Temperature and Pressure14m
- Dalton's Law: Partial Pressure (Simplified)13m
- Gas Stoichiometry18m
- Intermolecular Forces (Simplified)19m
- Intermolecular Forces and Physical Properties11m
- Atomic, Ionic and Molecular Solids10m
- Heating and Cooling Curves30m
- 9. Solutions4h 27m
- Solutions6m
- Solubility and Intermolecular Forces17m
- Solutions: Mass Percent6m
- Percent Concentrations10m
- Molarity18m
- Osmolarity15m
- Parts per Million (ppm)13m
- Solubility: Temperature Effect8m
- Intro to Henry's Law4m
- Henry's Law Calculations12m
- Dilutions12m
- Solution Stoichiometry14m
- Electrolytes (Simplified)13m
- Equivalents11m
- Molality15m
- The Colligative Properties15m
- Boiling Point Elevation16m
- Freezing Point Depression9m
- Osmosis16m
- Osmotic Pressure10m
- Vapor Pressure Lowering (Raoult's Law)16m
- 10. Acids and Bases3h 10m
- Acid-Base Introduction11m
- Arrhenius Acid and Base6m
- Bronsted Lowry Acid and Base21m
- Acid and Base Strength17m
- Ka and Kb16m
- The pH Scale16m
- Auto-Ionization9m
- pH of Strong Acids and Bases9m
- Acid-Base Equivalents14m
- Acid-Base Reactions7m
- Gas Evolution Equations (Simplified)6m
- Ionic Salts (Simplified)11m
- Buffers11m
- Henderson-Hasselbalch Equation16m
- Strong Acid Strong Base Titrations (Simplified)13m
- 11. Nuclear Chemistry1h 1m
- BONUS: Lab Techniques and Procedures1h 38m
- BONUS: Mathematical Operations and Functions47m
- 12. Introduction to Organic Chemistry1h 34m
- 13. Alkenes, Alkynes, and Aromatic Compounds2h 30m
- Spatial Orientation of Bonds3m
- Intro to Hydrocarbons16m
- Isomers14m
- Chirality15m
- Naming Alkenes11m
- Naming Dienes and Trienes6m
- Naming Alkynes9m
- Intro to Addition Reactions4m
- Halogenation Reaction4m
- Hydrogenation Reaction3m
- Hydrohalogenation Reaction7m
- Hydration Reaction10m
- Naming Benzene19m
- Benzene Reactions10m
- Benzene Reaction: Nitration6m
- Benzene Reaction: Sulfonation5m
- 14. Compounds with Oxygen or Sulfur1h 22m
- 15. Aldehydes and Ketones1h 1m
- 16. Carboxylic Acids and Their Derivatives1h 11m
- 17. Amines40m
- 18. Amino Acids and Proteins2h 2m
- 19. Enzymes1h 37m
- 20. Carbohydrates1h 46m
- Intro to Carbohydrates4m
- Classification of Carbohydrates4m
- Fischer Projections4m
- Enantiomers vs Diastereomers7m
- D vs L Enantiomers9m
- Cyclic Hemiacetals8m
- Intro to Haworth Projections4m
- Cyclic Structures of Monosaccharides11m
- Mutarotation4m
- Reduction of Monosaccharides10m
- Oxidation of Monosaccharides7m
- Glycosidic Linkage14m
- Disaccharides7m
- Polysaccharides8m
- 21. The Generation of Biochemical Energy2h 9m
- 22. Carbohydrate Metabolism2h 31m
- 23. Lipids2h 26m
- Intro to Lipids6m
- Fatty Acids25m
- Physical Properties of Fatty Acids6m
- Waxes4m
- Triacylglycerols12m
- Triacylglycerol Reactions: Hydrogenation8m
- Triacylglycerol Reactions: Hydrolysis13m
- Triacylglycerol Reactions: Oxidation7m
- Glycerophospholipids15m
- Sphingomyelins13m
- Steroids15m
- Cell Membranes7m
- Membrane Transport10m
- 24. Lipid Metabolism1h 45m
- 25. Protein and Amino Acid Metabolism1h 37m
- 26. Nucleic Acids and Protein Synthesis2h 54m
- Intro to Nucleic Acids4m
- Nitrogenous Bases16m
- Nucleoside and Nucleotide Formation9m
- Naming Nucleosides and Nucleotides13m
- Phosphodiester Bond Formation7m
- Primary Structure of Nucleic Acids11m
- Base Pairing10m
- DNA Double Helix6m
- Intro to DNA Replication20m
- Steps of DNA Replication11m
- Types of RNA10m
- Overview of Protein Synthesis4m
- Transcription: mRNA Synthesis9m
- Processing of pre-mRNA5m
- The Genetic Code6m
- Introduction to Translation7m
- Translation: Protein Synthesis18m
Naming Alcohols (Common Names): Videos & Practice Problems
Simple alcohols are named using common names that combine the alkyl group with the word "alcohol," such as methyl alcohol or propyl alcohol. This naming convention highlights the functional group characteristic of alcohols, which is essential in organic chemistry for identifying compounds. Understanding alkyl groups and their role in naming alcohols aids in grasping broader concepts like functional groups and molecular structure, which are foundational for studying reactions involving alcohols, such as oxidation or substitution. Mastery of these basics supports deeper learning in organic synthesis and biochemical pathways involving alcohols.
Naming Alcohols (Common Names) Concept 1
Naming Alcohols (Common Names) Concept 1 Video Summary
Naming Alcohols (Common Names) Example 1
Naming Alcohols (Common Names) Example 1 Video Summary
When naming alcohols using their common names, the key step is to identify the length of the alkyl group attached to the hydroxyl (–OH) group. For example, if the alcohol has a straight chain of four carbon atoms, the alkyl group is called "butyl." Since the compound contains an –OH group, the name ends with "alcohol." Therefore, a four-carbon straight-chain alcohol is commonly named butyl alcohol. This approach simplifies naming by focusing on the alkyl group and appending "alcohol" to indicate the presence of the hydroxyl functional group.
Provide common name for the following alcohol compound.
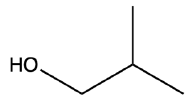
2-methyl-1-propanol.
Tert-butyl alcohol.
Isobutyl alcohol.
Propyl alcohol.
What is the common name for this alcohol?
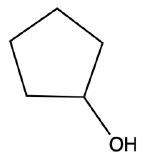
Pentyl alcohol.
Cyclohexyl alcohol.
Cyclopropyl alcohol.
Cyclopentyl alcohol.
Do you want more practice?
More setsGo over this topic definitions with flashcards
More setsHere's what students ask on this topic:
The common naming convention for simple alcohols involves combining the name of the alkyl group with the word "alcohol." This means you first identify the alkyl group attached to the hydroxyl (-OH) functional group, then add the word "alcohol" after it. For example, if the alkyl group is methyl (CH3-), the alcohol is called methyl alcohol. Similarly, propyl alcohol refers to an alcohol with a propyl group (C3H7-). This naming system is straightforward and highlights the functional group characteristic of alcohols, making it easier to recognize and communicate the structure of simple alcohols in organic chemistry.
To identify the alkyl group in an alcohol for common naming, first locate the carbon chain attached directly to the hydroxyl (-OH) group. This carbon chain is the alkyl group. For example, in CH3CH2OH, the alkyl group is ethyl (C2H5-). Once identified, the alkyl group's name is combined with the word "alcohol" to form the common name, such as ethyl alcohol. Recognizing the alkyl group is essential because it determines the prefix in the common name and helps distinguish different alcohols based on their carbon chain length and structure.
Understanding the common names of alcohols is important because it provides a foundational way to recognize and communicate the structure of alcohol compounds quickly. Common names emphasize the alkyl group attached to the hydroxyl functional group, which is crucial for identifying the molecule's properties and reactivity. This knowledge supports learning more complex topics such as oxidation, substitution reactions, and biochemical pathways involving alcohols. Additionally, mastering common names helps students transition to systematic IUPAC naming and understand how functional groups influence molecular behavior in organic synthesis.
Yes, here are some examples of common names for alcohols based on their alkyl groups:
1. Methyl alcohol (CH3OH) – the simplest alcohol with a methyl group.
2. Ethyl alcohol (C2H5OH), commonly known as ethanol.
3. Propyl alcohol (C3H7OH) – an alcohol with a propyl group.
4. Hexyl alcohol (C6H13OH) – an alcohol with a six-carbon alkyl chain.
These names follow the pattern of naming the alkyl group first, then adding "alcohol," which helps in quickly identifying the structure and size of the alcohol molecule.
The common naming system for alcohols names the compound by combining the alkyl group name with the word "alcohol," such as methyl alcohol or propyl alcohol. This system is simpler and often used for simple alcohols. In contrast, the IUPAC naming system is more systematic and precise. It names alcohols based on the longest carbon chain containing the hydroxyl group, replacing the '-e' ending of the alkane with '-ol' (e.g., methanol, ethanol, propanol). IUPAC names also include numbering to indicate the position of the hydroxyl group if necessary. While common names are easier for simple alcohols, IUPAC names provide clarity for more complex molecules.

