Textbook Question
What products (including stereoisomers, if applicable) are formed from the reaction of 3-bromo-3-methylpentane:
b. with H2O?
1179
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 9 - Substitution and Elimination Reactions of Alkyl Halides
Ch. 9 - Substitution and Elimination Reactions of Alkyl Halides Problem 64c
Problem 64c
 Verified step by step guidance
Verified step by step guidance
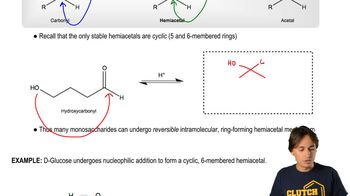


What products (including stereoisomers, if applicable) are formed from the reaction of 3-bromo-3-methylpentane:
b. with H2O?
Identify the three products formed when 2-bromo-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water.
Draw the products of the following intramolecular reactions:
a.
b.
Draw the products of the following intramolecular reactions:
e.
Draw the products of the following intramolecular reactions:
c.
d.
After a proton is removed from the OH group, which compound in each pair forms a cyclic ether more rapidly?
a.
b.