Textbook Question
For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them:
i.
j.
1084
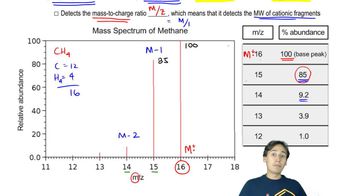
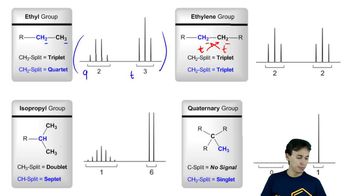
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 13 - Mass Spectrometry; Infrared Spectroscopy; UV/Vis Spectroscopy
Ch. 13 - Mass Spectrometry; Infrared Spectroscopy; UV/Vis Spectroscopy Problem 41
Problem 41 Verified step by step guidance
Verified step by step guidance

For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them:
i.
j.
At pH = 7, one of the ions shown here is purple and the other is blue. Which is which?
What would be the difference in the colors of the compounds at pH = 3?
For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them:
c. CH3CH2CH2OH and CH3CH2OCH3
d.
Rank each set of compounds in order of decreasing λmax:
b.
Rank the following compounds in order of increasing lmax: