Rank the following compounds from highest wavenumber to lowest wavenumber for their C=O absorption band:

 Bruice 8th Edition
Bruice 8th Edition Ch. 13 - Mass Spectrometry; Infrared Spectroscopy; UV/Vis Spectroscopy
Ch. 13 - Mass Spectrometry; Infrared Spectroscopy; UV/Vis Spectroscopy Problem 53
Problem 53What hydrocarbons that contain a six-membered ring will have a molecular ion peak at m/z = 112?
 Verified step by step guidance
Verified step by step guidance
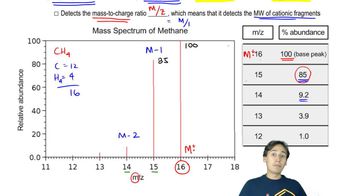
Verified video answer for a similar problem:
Key Concepts
Hydrocarbons

Mass Spectrometry
Cyclohexane Derivatives

A compound gives a mass spectrum with essentially only three peaks at m/z = 77 (40%), 112 (100%), and 114 (33%). Identify the compound.
Rank the following compounds from highest wavenumber to lowest wavenumber for their C-O absorption band:
Norlutin and Enovid are ketones that suppress ovulation, so they have been used clinically as contraceptives. For which of these compounds would you expect the infrared carbonyl absorption (C=O stretch) to be at a higher frequency? Explain.
The mass spectrum for a compound with molecular weight of 102 is shown below. Its IR spectrum has a broad, strong absorption at 3600 cm–1 and a medium absorption at 1360 cm–1.
a. Identify the compound.
<IMAGE>
A mass spectrum shows significant peaks at m/z = 87, 115, 140, and 143. Which of the following compounds is responsible for that mass spectrum?