Textbook Question
Identify the peaks in the mass spectrum of octan-4-one that correspond to (a) α-cleavage.
<IMAGE>
1060
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 14 - Structural Identification I: Infrared Spectroscopy and Mass Spectrometry
Ch. 14 - Structural Identification I: Infrared Spectroscopy and Mass Spectrometry Problem 45
Problem 45 Verified step by step guidance
Verified step by step guidance
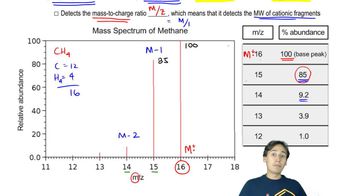
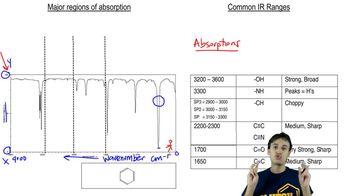

Identify the peaks in the mass spectrum of octan-4-one that correspond to (a) α-cleavage.
<IMAGE>
Identify the peaks in the mass spectrum of octan-4-one that correspond to (b) the McLafferty rearrangement.
<IMAGE>
Based on Hooke's law, choose the bond in each pair that you expect to vibrate at a higher wavenumber.
(f) C―S vs C=O
Based on Hooke's law, choose the bond in each pair that you expect to vibrate at a higher wavenumber.
(e) C=N vs C≡N
2-Methylnonan-4-one can undergo two different McLafferty rearrangements. Draw the products of each of them.
Choose the bond in each pair that you expect to have the more intense stretching band
(b) C=O vs. C=N