Textbook Question
For each of the molecules shown, predict the structure of at least one major fragment in the mass spectrum.
(d)
1103
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 14 - Structural Identification I: Infrared Spectroscopy and Mass Spectrometry
Ch. 14 - Structural Identification I: Infrared Spectroscopy and Mass Spectrometry Problem 64
Problem 64 Verified step by step guidance
Verified step by step guidance
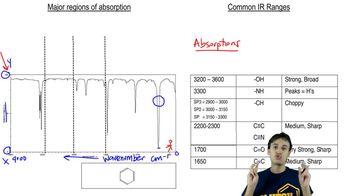
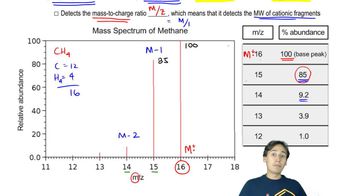

For each of the molecules shown, predict the structure of at least one major fragment in the mass spectrum.
(d)
For each of the molecules shown, predict the structure of at least one major fragment in the mass spectrum.
(e)
What ratio of M, M + 2 , M + 4 and would you expect for 1,6-dibromohexane?
For each of the molecules shown, predict the structure of at least one major fragment in the mass spectrum.
(b)