Textbook Question
In what direction is the dipole moment in calicene? Explain.
1412
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 8 - Delocalized Electrons: Their Effect on Stability, pKa, and the Products of a Reaction • Aromaticity and Electronic Effects: An Introduction to the Reactions of Benzene
Ch. 8 - Delocalized Electrons: Their Effect on Stability, pKa, and the Products of a Reaction • Aromaticity and Electronic Effects: An Introduction to the Reactions of Benzene Problem 52b
Problem 52b Verified step by step guidance
Verified step by step guidance

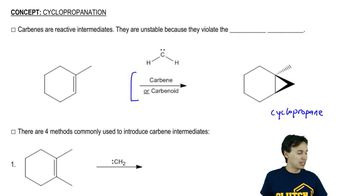

In what direction is the dipole moment in calicene? Explain.
Which compound in each set is aromatic? Explain your choice.
a.
Predict the relative pKa values of cyclopentadiene and cycloheptatriene.
In what direction is the dipole moment in fulvene? Explain.
Which compound in each set is aromatic? Explain your choice.
b.
Following the instructions for drawing the energy levels of the molecular orbitals for the compounds shown in [Figure 8.17], draw the energy levels of the molecular orbitals for the cycloheptatrienyl cation. For each compound, show the distribution of the electrons. Which of the compounds are aromatic?