Textbook Question
Predict the hybridization and geometry of the carbon and nitrogen atoms in the following molecules and ions. (Hint: Resonance.)
d.
1052
views

 Verified step by step guidance
Verified step by step guidance
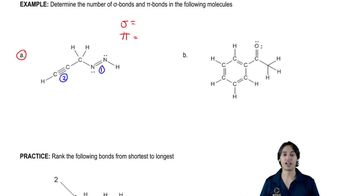
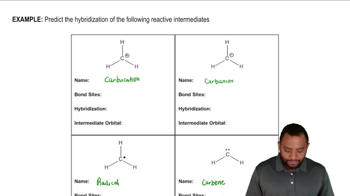
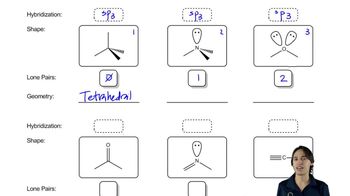
Predict the hybridization and geometry of the carbon and nitrogen atoms in the following molecules and ions. (Hint: Resonance.)
d.
a. Draw the structure of cis-CH3CH=CHCH2CH3 showing the pi bond with its proper geometry.
b. Circle the six coplanar atoms in this compound.
Predict the hybridization and geometry of the carbon and nitrogen atoms in the following molecules and ions. (Hint: Resonance.)
f.
Draw orbital pictures of the pi bonding in the following compounds:
f. CH3CH=NCH=C=O
Draw orbital pictures of the pi bonding in the following compounds:
e. CH3CH=C=CHCH3
Predict the hybridization and geometry of the carbon and nitrogen atoms in the following molecules and ions. (Hint: Resonance.)
e.