For each of the following reactions, identify the bonds that are broken and formed. Be sure to indicate whether the bond that is broken is a σ bond or a π bond.
(a)

 Mullins 1st Edition
Mullins 1st Edition Ch. 5 - Chemical Reaction Analysis: Thermodynamics and Kinetics
Ch. 5 - Chemical Reaction Analysis: Thermodynamics and Kinetics Problem 49c
Problem 49c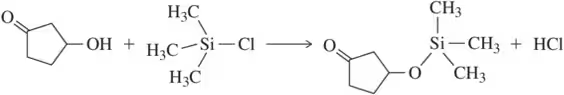
 Verified step by step guidance
Verified step by step guidance

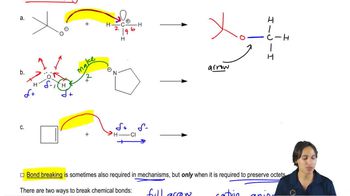

For each of the following reactions, identify the bonds that are broken and formed. Be sure to indicate whether the bond that is broken is a σ bond or a π bond.
(a)
For each of the following reactions, identify the bonds that are broken and formed. Be sure to indicate whether the bond that is broken is a σ bond or a π bond.
(e)
The A value of a substituent on a cyclohexane ring is essentially the ∆G° for a substituent going from the equatorial to the axial position in a chair–chair interconversion. Because most substituents prefer to be in the equatorial position, A values are, by definition, positive numbers. Use the table of A values to calculate ∆G° and Keq for the chair–chair interconversions shown.
(b)
For each of the following acid–base reactions, (ii) calculate Keq. If a pKa is not one of the ten common ones we learned in Chapter 4, it will be given to you.
(a)
For each of the following acid–base reactions, (iii) calculate ∆G°. If a pKa is not one of the ten common ones we learned in Chapter 4, it will be given to you.
(a)
The A value of a substituent on a cyclohexane ring is essentially the ∆G° for a substituent going from the equatorial to the axial position in a chair–chair interconversion. Because most substituents prefer to be in the equatorial position, A values are, by definition, positive numbers. Use the table of A values to calculate ∆G° and Keq for the chair–chair interconversions shown.
(a)